In 2017, researchers at Children’s Hospital of Philadelphia made waves when they revealed an artificial womb that could support premature lambs, giving their fragile lungs and brains more time to develop. Now, the team is seeking approval for the first human trials of this experimental technology.
The artificial womb, called the Extra-uterine Environment for Newborn Development (EXTEND), aims to improve outcomes for extremely premature babies born between 22-28 weeks gestation. At this stage, infants often struggle to survive outside the womb and frequently face lifelong disabilities.
EXTEND works by placing the premature baby into a sterile bag filled with a fluid that mimics amniotic fluid. Surgeons connect the umbilical cord to a machine that pumps blood through an artificial placenta, providing oxygen and removing waste. The goal is to give the infant’s vital organs crucial extra time to mature before facing the outside world.

In their 2017 study, the researchers sustained lambs equivalent to 23 weeks human gestation for up to four weeks. The animals grew wool, their lungs expanded, and their brains showed advanced development.
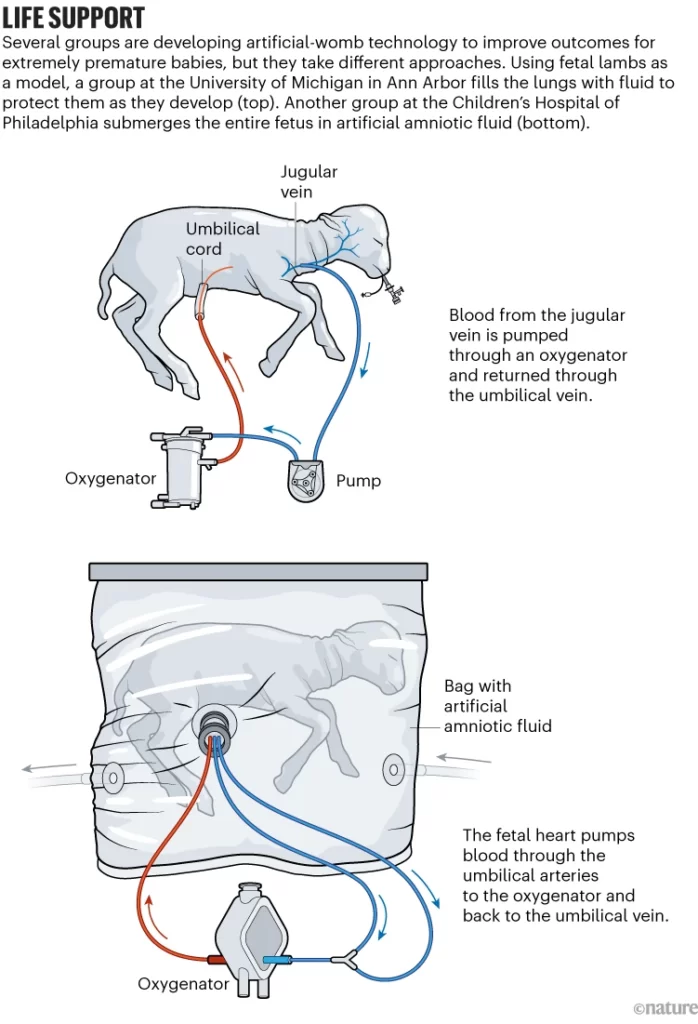
Other groups around the world are also racing to develop similar artificial womb technologies. But bioethicists warn we still need more data before human trials begin. Lambs are quite different from humans at the same stage of fetal development. Successful experiments in non-human primates would provide more assurance of safety and efficacy.
There are also wider concerns around the ethics and accessibility of artificial wombs. Some fear the technology could one day threaten natural pregnancy, but experts say that prospect is science fiction, not science. Others worry artificial wombs could alter the viability timeframe for legal abortion. And many caution that focusing on a high-tech solution ignores deeper inequities around why women deliver prematurely in the first place.
Still, the need is urgent for better care of the world’s 13.4 million premature infants. If artificial wombs can buy these fragile babies vital time, many feel the promise outweighs the uncertainties. All eyes will be on September’s FDA meeting as we await the next chapter in this story.
References
- Partridge, E. A. et al. Nature Commun. 8, 15112 (2017).Article PubMed Google Scholar
- Crump, C., Winkleby, M. A., Sundquist, J. & Sundquist, K. JAMA 322, 1580–1588 (2019).Article PubMed Google Scholar
- Flake, A. W., De Bie, F. R., Munson, D. A. & Feudtner, C. J. Perinatol. https://doi.org/10.1038/s41372-023-01716-2 (2023).
- Kukora, S. K., Mychaliska, G. B. & Weiss, E. M. J. Perinatol. https://doi.org/10.1038/s41372-023-01713-5 (2023).
- McLeod, J. S. et al. J. Pediatr. Surg. 54, 1147–1152 (2019).